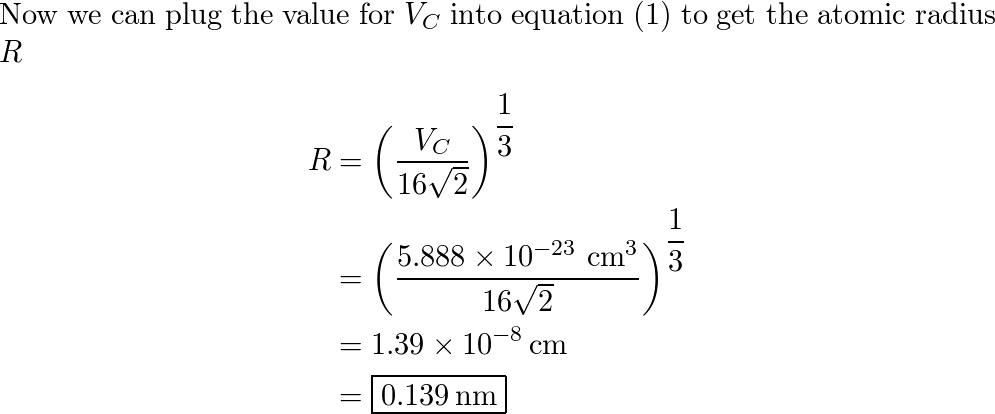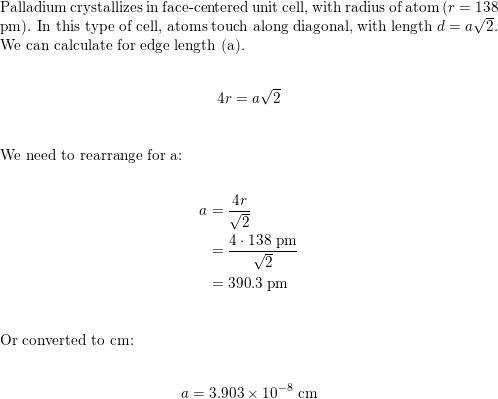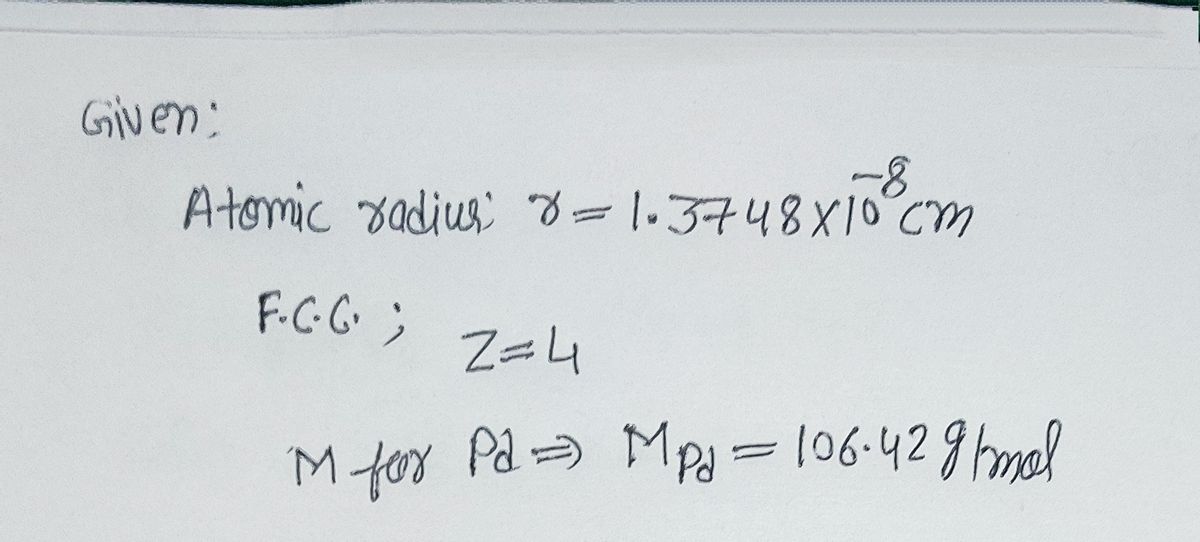
A mineral having the formula AB2 crystallizes in the cubic closepacked lattice with the A atoms occupying the lattice points What are the coordination numbers of the A and B atoms

Gold occurs as face centred cube and it has a density of 19.30 kg dm ^-3 .Calculate atomic radius of gold. (Molar mass of Au = 197 )

SOLVED: A metal crystallizes in the face-centered cubic (FCC) lattice. The density of the metal is 8902 kglm , and the length of a unit cell edge, a,is 352.4 pm. Calculate the
An element crystallizes in an FCC lattice and the edge of the unit cell is 0.559nm. The density is 3.19g/cm. What is the atomic weight? - Quora

SOLVED: Rhodium has a density of 12.41 g>cm3 and crystallizes with the face-centered cubic unit cell. Calculate the radius of a rhodium atom.

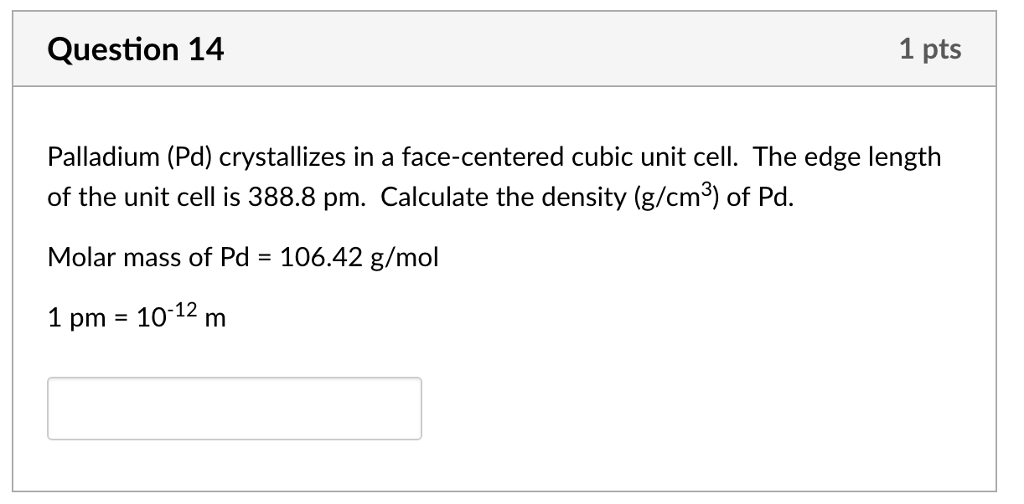
OneClass: Palladium crystallizes with a face-centered cubic structure. It hasa density of 12.0 g/cm3,...
PLEASE HELP! 80 points!! A metal crystallizes in the face‑centered cubic (FCC) lattice. The density of the - Brainly.com
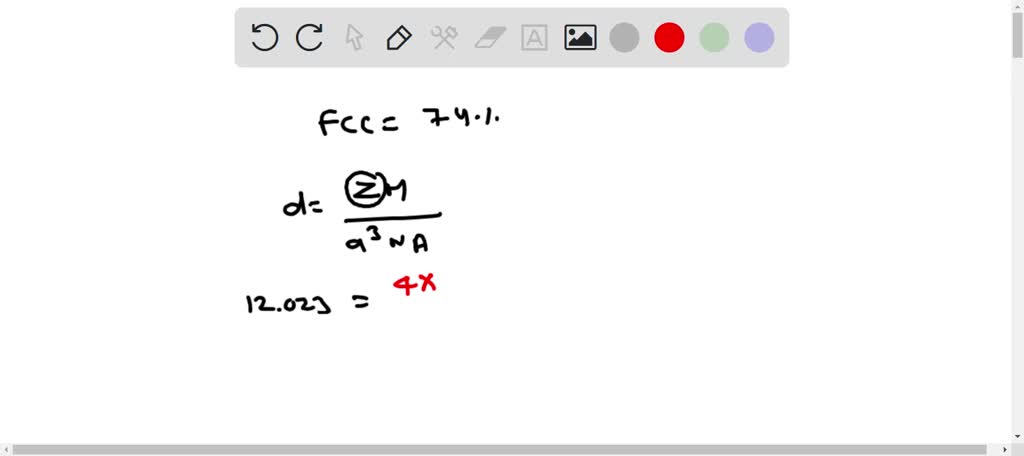
SOLVED: Palladium (at. wt. = 106) crystallizes in a face-centered cubic unit cell. Its density is 12.023 g/cm3 . Calculate the atomic radius of palladium and its packing efficiency.

OneClass: A metal crystallizes in the face-centered cubic (FCC) lattice. The density of the metal is ...

Problem.docx - Problem #1: Palladium crystallizes in a face-centered cubic unit cell. Its density is 12.023 g/cm3. Calculate the atomic radius | Course Hero

A metal crystallizes in the face-centered cubic unit cell with an edge length of 320 pm. \\ A. What is the radius of the metal atom? B. The density of the metal

Gold occurs as face centred cube and it has a density of 19.30 kg dm ^-3 .Calculate atomic radius of gold. (Molar mass of Au = 197 )

Interactions between Hydrogen and Palladium Nanoparticles: Resolving Adsorption and Absorption Contributions - Moumaneix - 2023 - ChemElectroChem - Wiley Online Library